UCLA Scientists Uncover "Zombie" Immune Cells Driving Aging and Fatty Liver Disease, Paving Way for Novel Therapeutic Strategies
UCLA scientists have made a significant breakthrough, identifying a previously overlooked yet highly detrimental group of immune cells that stealthily accumulates in aging tissues and, notably, in the livers of individuals afflicted with fatty liver disease. The removal of these specific cells in murine models resulted in a dramatic reduction in inflammation and a reversal of liver damage, a remarkable outcome achieved even while the animals maintained an unhealthy dietary regimen. This discovery not only sheds new light on the mechanisms underlying chronic liver disease and aging but also opens promising avenues for the development of innovative therapeutic interventions.
Understanding Cellular Senescence: The "Zombie Cell" Phenomenon
The research, published in the esteemed journal Nature Aging, centers on cellular senescence, a complex biological process initiated by various forms of cellular stress. In this state, cells cease to divide but do not undergo programmed cell death (apoptosis). Instead, these persistent cells, colloquially termed "zombie cells," remain metabolically active within tissues, continuously secreting a cocktail of pro-inflammatory signals. This steady stream of inflammatory mediators, collectively known as the Senescence-Associated Secretory Phenotype (SASP), can profoundly damage surrounding healthy cells and contribute to tissue dysfunction and chronic disease.
Cellular senescence has long been implicated in the aging process and a wide array of age-related pathologies, including cardiovascular disease, neurodegenerative disorders, metabolic syndromes, and certain cancers. The accumulation of senescent cells is a hallmark of biological aging, contributing to the chronic low-grade inflammation often observed in older individuals, known as "inflammaging." This chronic inflammation is a key driver of age-related decline and disease progression.
"Senescent cells are fairly rare, but think of them like a broken-down car on the 405," explained Anthony Covarrubias, senior author of the study and a distinguished member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. "Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption." This vivid analogy underscores the disproportionate impact a small population of dysfunctional cells can have on an entire organ system.
Unmasking the Culprits: The Macrophage Mystery Solved
For many years, the scientific community debated whether macrophages, the versatile immune cells renowned for their "scavenger" role in patrolling the body and clearing cellular debris, could truly become senescent. A significant portion of researchers held the belief that macrophages were exempt from this process. This skepticism stemmed from the inherent difficulty in distinguishing between healthy macrophages, which naturally exhibit some molecular features reminiscent of senescent cells, and macrophages that had indeed entered a dysfunctional, senescent state. The overlapping molecular profiles made clear identification a considerable challenge.
The UCLA team meticulously addressed this critical problem by painstakingly identifying a definitive molecular signature for senescent macrophages. Their breakthrough involved pinpointing the concurrent expression of two specific proteins: p21 and TREM2. This unique combination was found to reliably mark macrophages that were genuinely senescent, no longer performing their essential functions effectively, yet paradoxically still actively driving inflammation in adjacent tissues. The identification of this clear biomarker is a pivotal step, providing researchers with an unequivocal tool to study these elusive cells.
Utilizing this novel p21-TREM2 marker, the researchers observed a dramatic and concerning shift in macrophage populations correlated with age. In young, healthy mice, senescent macrophages constituted only approximately 5% of the total liver macrophage population. However, in older mice, this figure surged dramatically, reaching an alarming 60% to 80%. This substantial age-related increase in senescent macrophages in the liver closely mirrored the observed rise in chronic liver inflammation that is a characteristic feature of aging. This strong correlation provided compelling evidence for the direct involvement of these senescent immune cells in age-related liver pathology.
The Cholesterol Connection: A Potent Trigger for Senescence
While aging is an undeniable factor in the accumulation of senescent cells, the UCLA team’s investigation unveiled another critical trigger: excess cholesterol. Their findings demonstrated that elevated levels of cholesterol can actively push macrophages into a senescent state. When healthy macrophages were exposed to high concentrations of low-density lipoprotein (LDL) cholesterol – often referred to as "bad" cholesterol – in laboratory settings, they exhibited a distinct transformation. These cells ceased their normal division, began secreting pro-inflammatory proteins, and, crucially, displayed the characteristic p21-TREM2 signature, confirming their transition into senescence.
"Physiologically, macrophages are well-equipped to handle cholesterol metabolism," noted Ivan Salladay-Perez, first author of the new study and a graduate student in the Covarrubias lab. "But in a chronic state, it becomes pathological. And when you look at fatty liver disease, which is fundamentally driven by overnutrition and excessive cholesterol in the blood, that surplus cholesterol appears to be a major driver of the senescent macrophage population." This insight provides a direct molecular link between diet, cholesterol levels, and the onset of cellular senescence, particularly in the context of metabolic diseases.
This critical discovery raises a broader and more profound possibility: that diets persistently high in fat and cholesterol may not only contribute to specific organ damage but could also accelerate biological aging across multiple organ systems. By promoting macrophage senescence, such diets might contribute to the aging process not only in the liver but potentially also in other vital organs like the brain, where microglia (brain macrophages) play a key role, the heart and vascular system (contributing to atherosclerosis), and even in adipose (fat) tissue, which is itself a metabolically active organ. This suggests a systemic impact of dietary choices on the aging trajectory.
Dramatic Reversal: Clearing Senescent Cells Reverses Liver Damage
To definitively test the hypothesis that removing these senescent cells could lead to tangible health improvements, the research team administered ABT-263, a known senolytic drug designed to selectively induce apoptosis (programmed cell death) in senescent cells, to their mouse models. The therapeutic effects observed were nothing short of dramatic. In mice that had been maintained on a high-fat, high-cholesterol diet – a regimen known to induce fatty liver disease – treatment with ABT-263 led to a significant reduction in liver size. Liver mass dropped from approximately 7% of total body weight to a much healthier range of 4-5%. Concurrently, the overall body weight of the treated animals also experienced a notable decrease of about 25%, falling from roughly 40 grams to around 30 grams.
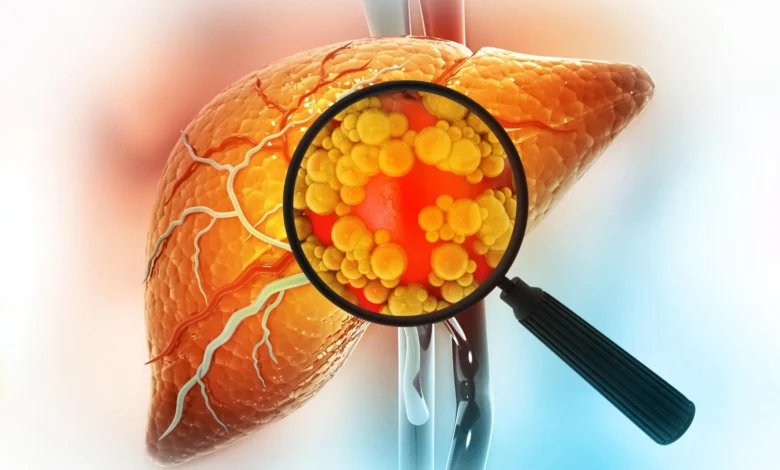
Visually, the treated livers presented a stark contrast to those of untreated animals. They appeared smaller, healthier, and exhibited a normal reddish coloration, indicative of healthy tissue. In comparison, the livers from untreated mice on the same unhealthy diet were visibly enlarged, often discolored with a yellowish hue, characteristic of severe fat accumulation and damage.
These compelling results strongly suggest that the selective elimination of senescent macrophages alone can yield substantial metabolic improvements, even in the absence of any dietary modifications. "That’s what wowed me," Salladay-Perez remarked, highlighting the unexpected potency of the intervention. "Eliminating senescent cells doesn’t just slow the fatty liver — it actually reverses it." This finding challenges conventional wisdom, suggesting that targeting underlying cellular mechanisms could offer a powerful therapeutic strategy independent of direct lifestyle changes, though the latter remain crucial for prevention.
Human Relevance: A Pressing Public Health Imperative
To ascertain the applicability of these groundbreaking findings to human health, the researchers meticulously analyzed an existing genomic dataset derived from human liver biopsies. Their investigation revealed a critical parallel: the same distinct senescent macrophage signature identified in mice was significantly more prevalent and pronounced in diseased human livers compared to healthy ones. This robust correlation provides compelling evidence that macrophage senescence is likely a significant contributor to the pathogenesis and progression of chronic liver disease in human populations.
The implications of this finding are particularly urgent given the escalating global burden of fatty liver disease, specifically Non-Alcoholic Fatty Liver Disease (NAFLD) and its more severe form, Non-Alcoholic Steatohepatitis (NASH). NAFLD affects an estimated 25-30% of the global adult population, with prevalence rates even higher in developed nations and among specific ethnic groups. In the United States, NAFLD affects approximately 80 to 100 million people, and NASH, which can progress to cirrhosis, liver failure, and hepatocellular carcinoma, affects 16 million Americans. The issue is especially pressing in metropolitan areas like Los Angeles, where an estimated 30-40% of residents are affected by fatty liver disease, with disproportionately higher rates observed in Latino communities. This demographic disparity underscores the complex interplay of genetic, environmental, and socio-economic factors in disease susceptibility.
Current treatment options for NAFLD and NASH remain remarkably limited, primarily revolving around lifestyle modifications such as diet and exercise, which can be challenging for many patients to sustain. Furthermore, effective early detection tools that can accurately identify individuals at risk of progression to severe liver damage are still lacking. This therapeutic void makes the UCLA team’s discovery particularly impactful, offering a potential new pharmacological approach to a widespread and debilitating condition.
"This is a huge public health crisis in the making," stated Covarrubias, who is also an assistant professor of microbiology, immunology, and molecular genetics. "We’re seeing fatty liver disease in younger and younger people. So we’re really happy to make some inroads into understanding what’s driving it and identifying cell types we might be able to target." The increasing incidence of fatty liver disease in younger demographics signals a looming health catastrophe, making research into fundamental drivers and targeted interventions more critical than ever.
Towards New Therapies: The Road Ahead for Senolytics
While the senolytic drug ABT-263 demonstrated profound therapeutic efficacy in murine models, its clinical application in humans is currently hampered by significant toxicity concerns, primarily dose-limiting myelosuppression. This challenge necessitates a concerted effort to identify safer and more selectively targeted compounds. The UCLA research team is keenly aware of this hurdle and plans to embark on comprehensive screening initiatives to discover novel senolytic agents. The goal is to identify compounds that can selectively eliminate senescent macrophages without eliciting harmful side effects in human patients, thus paving the way for a new class of precision medicines.
The pharmaceutical industry has shown growing interest in senolytics, with several companies actively engaged in drug discovery and development. The success of ABT-263 in a preclinical setting underscores the potential of this therapeutic strategy and will likely spur further investment and research into this promising field. The eventual goal is to develop orally available drugs that can safely and effectively clear senescent cells, thereby preventing or reversing age-related diseases.
Broader Horizons: The Geroscience Hypothesis and Systemic Impact
These findings lend powerful support to the geroscience hypothesis, a burgeoning paradigm in medical research that posits that the fundamental biological processes of aging are major drivers of multiple chronic diseases. This hypothesis suggests that by targeting these shared underlying mechanisms of aging, it may be possible to simultaneously prevent or treat a broad spectrum of age-related conditions, rather than addressing each disease in isolation.
In this context, the accumulation of senescent macrophages emerges as a critical common denominator. Their persistent inflammatory signaling could contribute to a diverse range of conditions, extending far beyond fatty liver disease. This includes atherosclerosis, where chronic inflammation plays a pivotal role in plaque formation; Alzheimer’s disease and other neurodegenerative disorders, where microglia (the brain’s resident macrophages) are known to become dysfunctional and inflammatory; and even various forms of cancer, where senescent cells can create a pro-tumorigenic microenvironment.
The research team is already investigating whether similar processes involving senescent immune cells occur in other age-related diseases. For instance, in the brain, microglia are constantly encountering and clearing cellular debris. Under chronic stress or in the presence of excessive waste products, as seen in neurodegenerative conditions like Alzheimer’s disease, these critical immune cells may themselves become senescent, further exacerbating neural inflammation and damage.
"If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s and cancer," emphasized Salladay-Perez. "It all goes back to understanding how these cells arise in the first place." This holistic perspective underscores the transformative potential of geroscience, suggesting a paradigm shift from treating individual diseases to tackling the root causes of aging itself.
The study, a testament to collaborative scientific endeavor, received vital support from prestigious institutions, including the National Institutes of Health, the Glenn Foundation for Medical Research, the American Federation for Aging Research, and the UCLA-UCSD Diabetes Research Center. This collective backing highlights the broad recognition of the importance and potential impact of this groundbreaking research. The identification of senescent macrophages as a key driver of both aging and specific diseases offers a compelling new target for therapeutic development, holding immense promise for improving human health and extending healthy lifespans in the decades to come.