FDA Grants 510k Clearance to Zeto New Wave EEG System for Outpatient and Home Diagnostic Use
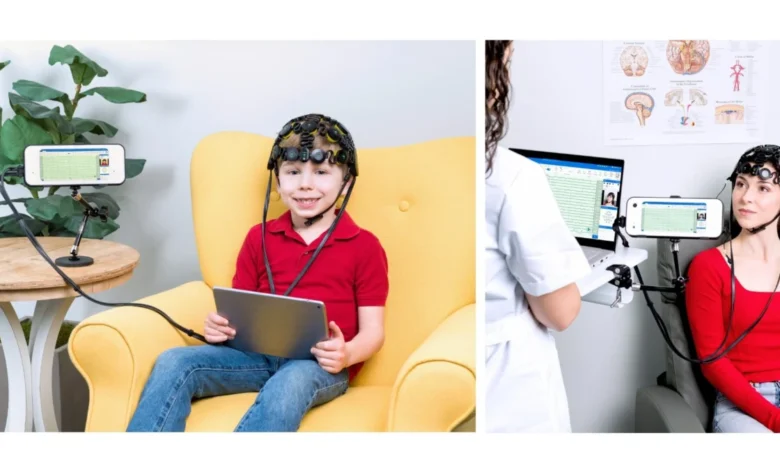
The United States Food and Drug Administration (FDA) has officially granted 510(k) clearance to Zeto Inc. for its latest medical innovation, the Zeto New Wave electroencephalogram (EEG) system. This regulatory milestone marks a significant advancement in the field of neurodiagnostics, specifically targeting the operational needs of outpatient clinics and the growing demand for home-based diagnostic capabilities. By integrating a sophisticated 21-electrode configuration with seamless cloud connectivity, the Zeto New Wave is positioned to mitigate the chronic staffing shortages currently plaguing the American healthcare system while simultaneously expanding patient access to essential neurological monitoring.
For years, the field of neurology has struggled with a logistical bottleneck: the traditional EEG setup. Conventional systems often require specialized technicians to manually apply conductive paste and position individual electrodes, a process that is both time-consuming and prone to human error. Furthermore, the scarcity of registered EEG technologists has led to significant wait times for patients, often stretching into several weeks or even months. The Zeto New Wave seeks to disrupt this paradigm by offering a streamlined, user-friendly alternative that does not compromise the clinical integrity of the data collected.
Technological Specifications and Design Innovation
The Zeto New Wave system is engineered to provide full-head coverage, utilizing 21 soft-tip, "no-mess" electrodes. These electrodes are strategically placed in accordance with the International 10-20 system, which is the globally recognized standard for electrode placement in clinical EEG. Unlike traditional "wet" EEG systems that require messy gels or pastes that must be washed out of the patient’s hair following a session, the New Wave’s dry-electrode approach enhances the patient experience by maintaining cleanliness and comfort.
The hardware component of the system includes a recording device characterized by an integrated display and intuitive manual controls. This allows healthcare providers to monitor signal quality in real-time without needing to toggle between multiple screens or external monitors. Beyond the standard brainwave activity, the device is capable of capturing synchronized high-definition video and audio. This synchronization is critical for neurologists when correlating physical movements or seizures with electrical spikes in the brain.
In addition to standard EEG data, the New Wave system offers modularity through the capture of optional physiological signals. These include electrocardiogram (ECG) for heart rate monitoring, electrooculogram (EOG) to track eye movements, and electromyogram (EMG) to monitor muscle activity. The inclusion of orientation sensors and external triggers for event-related potentials (ERPs) further broadens the diagnostic utility of the device. Notably, the system also features integrated photic stimulation detection, a necessary component for identifying photosensitive epilepsy and other reflexive neurological conditions.
Addressing the Clinical Staffing Crisis
The timing of the FDA clearance for the Zeto New Wave is particularly relevant given the current labor market in healthcare. According to data from the American Society of Electroneurodiagnostic Technologists (ASET), there is a growing disparity between the number of qualified EEG technologists and the increasing volume of patients requiring neurological screening. This shortage is exacerbated by the aging U.S. population and the rising prevalence of neurological disorders such as epilepsy, dementia, and various sleep-related conditions.
By simplifying the application process, the Zeto New Wave allows outpatient clinics to utilize a broader range of clinical staff to initiate EEG recordings. Because the headset is designed for ease of use, the requirement for a highly specialized technologist to be physically present for every routine setup is reduced. This shift enables clinics to increase their daily throughput, effectively reducing the backlog of patients waiting for diagnostic answers.
The system is optimized for short-term recordings, typically lasting up to 2.5 hours. This duration is ideal for routine clinical EEGs, which are the most common form of brainwave testing used to evaluate patients for seizure disorders or unexplained loss of consciousness. By focusing on this specific window of time, Zeto has created a tool that fits perfectly into the workflow of a busy physician’s practice or a specialized outpatient center.
Cloud Connectivity and the Digital Neurology Ecosystem
A cornerstone of Zeto’s technological philosophy is its reliance on a robust cloud platform. The Zeto New Wave operates on the proprietary Zeto Cloud, a secure, HIPAA-compliant environment that facilitates the immediate upload and storage of diagnostic data. This connectivity allows for a decentralized model of care where the data can be recorded in an outpatient clinic or a patient’s home and then reviewed remotely by a board-certified neurologist located anywhere in the world.
This remote access is a game-changer for rural healthcare facilities that may not have a neurologist on-site. It also streamlines the workflow for large hospital networks, allowing their specialists to interpret tests from multiple satellite clinics through a single interface. The Zeto Cloud also supports advanced quantitative analytics. Zeto has developed artificial intelligence (AI) tools that provide seizure detection and seizure burden analysis. These AI-powered insights act as a "second pair of eyes" for the physician, highlighting areas of concern within the EEG data and supporting faster, more accurate clinical decision-making.
Evolution of the Zeto Product Portfolio
The New Wave represents the third FDA-cleared device in Zeto’s portfolio, following the successful launches of the Zeto WR19 and the Zeto ONE. This progression demonstrates a clear strategic trajectory for the company:
- Zeto WR19: The first-generation device that proved the viability of the dry-electrode, wireless headset concept in a clinical setting.
- Zeto ONE: Released earlier in 2024, this system was specifically tailored for high-acuity environments, including Intensive Care Units (ICUs), Emergency Departments (EDs), and during patient transport. It addressed the need for rapid "stat" EEGs where time is of the essence.
- Zeto New Wave: The latest iteration, refined based on feedback from the WR19, designed specifically for the outpatient market and physician practices where patient volume and ease of use are the primary drivers.
Florian Strelzyk, PhD, Chief Executive Officer of Zeto, emphasized this evolutionary process in a recent statement. He noted that the New Wave was "built on lessons learned" from the first-generation WR19. Strelzyk highlighted that while the Zeto ONE serves the urgent needs of the ICU, the New Wave is the "ideal solution for physician practices," allowing providers to select a system that matches their specific clinical needs and budgetary constraints.
Clinical and Economic Implications
The broader implications of the Zeto New Wave clearance extend to the economics of neurology practices. Traditional EEG equipment involves high upfront costs, specialized room requirements (such as electrical shielding), and the ongoing expense of consumables and specialized labor. The Zeto system’s portability and cloud-based architecture reduce the need for dedicated EEG suites, allowing any exam room to be converted into a diagnostic space.
From a clinical perspective, the ability to perform high-quality EEGs in a home setting or an outpatient clinic can significantly improve the "diagnostic yield" for patients with infrequent symptoms. Many patients suffer from "white coat" effects or find it difficult to replicate their symptoms in a sterile hospital environment. By bringing the technology to the patient, rather than forcing the patient to the technology, the likelihood of capturing a transient neurological event increases.
Furthermore, the integration of ECG, EMG, and EOG signals into a single, synchronized recording allows for a more holistic view of the patient’s physiological state. For example, a neurologist can more easily distinguish between a primary cardiac event causing a syncopal episode and a primary neurological seizure.
Future Outlook: AI and Decentralized Care
As the healthcare industry continues to move toward a model of "hospital-at-home" and decentralized diagnostics, tools like the Zeto New Wave are likely to become the new standard. The convergence of wearable medical hardware, cloud computing, and artificial intelligence is creating a landscape where neurological monitoring is no longer a rare, high-effort event, but a streamlined part of routine preventative and diagnostic care.
Zeto’s commitment to AI-driven analytics also suggests a future where the initial screening of EEG data is automated, allowing neurologists to focus their expertise on the most complex cases. As seizure detection algorithms become more refined through machine learning, the speed of diagnosis for conditions like epilepsy will likely decrease, leading to faster initiation of life-changing treatments and medications.
In conclusion, the FDA 510(k) clearance of the Zeto New Wave is a pivotal moment for Zeto Inc. and the neurology community at large. By addressing the practical barriers of mess, staffing, and location, the system promises to democratize access to high-quality brain monitoring. As clinics across the United States adopt this new wave of technology, the ultimate beneficiaries will be the patients who receive faster, more comfortable, and more accurate neurological care.








