Johns Hopkins Researchers Identify CSE Protein and Hydrogen Sulfide as Critical Factors in Slowing Alzheimers Progression
Scientists at Johns Hopkins Medicine, supported by a significant new grant from the National Institutes of Health (NIH), have released findings that could redefine the therapeutic landscape for Alzheimer’s disease. The research centers on an enzyme called cystathionine γ-lyase, commonly referred to as CSE, and its role in producing hydrogen sulfide ($H_2S$). While hydrogen sulfide is widely recognized by the public for its distinctive "rotten egg" odor and its toxicity at high concentrations, this study highlights its indispensable role as a signaling molecule that protects the brain from the ravages of neurodegeneration.
The study, published in the Proceedings of the National Academy of Sciences (PNAS), suggests that maintaining precise, microscopic levels of hydrogen sulfide in the brain is essential for cognitive health. Led by Bindu Paul, M.S., Ph.D., an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, the research team utilized genetically engineered mice to demonstrate that the absence of CSE leads to a rapid decline in memory and learning, mirroring the progression of Alzheimer’s disease in humans.
The Biological Paradox of Hydrogen Sulfide
Hydrogen sulfide has long been a subject of interest in toxicology, where it is treated as a hazardous gas. However, in the realm of mammalian biology, it belongs to a class of molecules known as gasotransmitters. Like nitric oxide and carbon monoxide, hydrogen sulfide is produced endogenously—meaning within the body—to facilitate communication between cells and regulate various physiological processes.
In the brain, the CSE enzyme is the primary architect of this gas. The challenge for modern medicine has been the "Goldilocks" nature of $H_2S$: too much is lethal, but too little appears to leave the brain vulnerable to oxidative stress and structural decay. The Johns Hopkins team focused on understanding how the body naturally regulates these trace amounts and what happens when the production machinery fails.
The researchers discovered that CSE acts as a guardian of the brain’s cellular integrity. When CSE levels are sufficient, hydrogen sulfide helps neutralize free radicals—unstable molecules that damage DNA and proteins. Without it, the brain enters a state of high oxidative stress, a condition that has been linked to the accumulation of the amyloid-beta plaques and tau tangles characteristic of Alzheimer’s.
A Chronology of Discovery: From Blood Pressure to Brain Health
The latest findings are the culmination of nearly two decades of research involving several high-profile laboratories. To understand the significance of the current study, one must look at the timeline of how CSE and $H_2S$ have been perceived in the scientific community.
The journey began in 2008, when researchers first developed a strain of mice lacking the CSE protein. At that time, the focus was not on the brain, but on the cardiovascular system. Scientists discovered that CSE was a major regulator of blood vessel function and blood pressure, marking the enzyme as a critical player in heart health.
By 2014, the scope of research shifted toward neurology. A team led by Solomon Snyder, M.D., D.Sc., D.Phil.—a legendary figure in neuroscience and professor emeritus at Johns Hopkins—reported that CSE appeared to support brain health in models of Huntington’s disease. This was a landmark moment, as it suggested that the gasotransmitter’s protective qualities extended to the central nervous system.
In 2021, the group moved closer to the "Holy Grail" of neurodegeneration: Alzheimer’s disease. They observed that in mice specifically bred to develop Alzheimer’s symptoms, CSE activity was significantly diminished. Crucially, they found that administering very small, controlled injections of hydrogen sulfide could restore some degree of cognitive function.
The current 2024 study represents the definitive isolation of CSE’s role. Unlike previous experiments that used mice with multiple genetic mutations, this research looked at mice where only the CSE gene was deleted. This allowed the team to prove that the loss of CSE alone is enough to trigger an Alzheimer’s-like state, regardless of other genetic predispositions.
Experimental Evidence: The Barnes Maze and Cognitive Decline
To quantify the impact of CSE deficiency, the Johns Hopkins team employed the Barnes maze, a standard tool in behavioral neuroscience used to measure spatial learning and memory. The maze consists of a circular platform with multiple holes around the perimeter; only one hole leads to a dark, safe "escape box," while the others are shallow and offer no refuge from the bright lights and open space that mice find stressful.
The results were stark and age-dependent. At two months of age, the mice lacking the CSE protein performed indistinguishably from their healthy counterparts. Both groups were able to navigate the maze and find the escape box in roughly three minutes, suggesting that early development was not significantly hampered by the lack of the enzyme.
However, as the mice reached the six-month mark—roughly equivalent to middle age in humans—the CSE-deficient mice showed a dramatic decline. While the control mice continued to find the escape route quickly, the mice lacking CSE struggled with the task, exhibiting significant memory impairment.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute to CSE loss," noted Suwarna Chakraborty, a researcher in Paul’s lab and the first author of the study. This progression mirrors the human experience of Alzheimer’s, where symptoms often remain dormant for years before emerging in later stages of life.
Structural Decay and the Blood-Brain Barrier
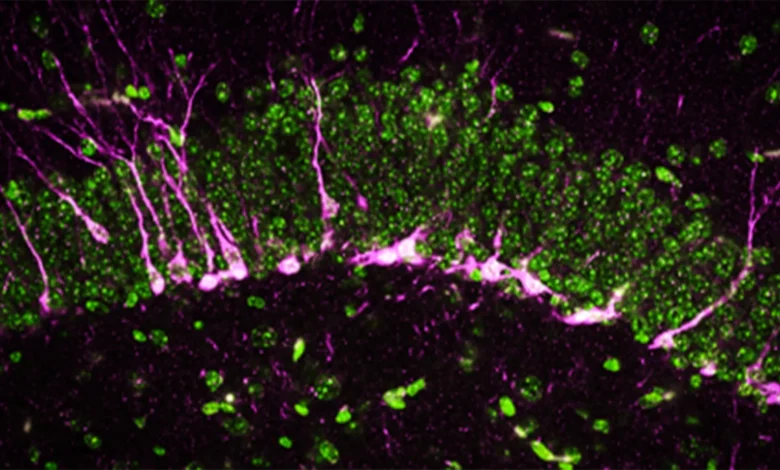
Beyond behavioral changes, the researchers used high-powered electron microscopy and biochemical analysis to look at the physical changes within the brain. They focused on the hippocampus, the region responsible for converting short-term memories into long-term ones.
One of the most significant findings involved neurogenesis—the birth of new neurons. In a healthy brain, the hippocampus constantly generates new cells that migrate to specific areas to support memory formation. In the CSE-deficient mice, the proteins required for this process were either missing or severely reduced. Furthermore, the few new neurons that were produced struggled to reach their destination, effectively "getting lost" in the brain’s architecture.
The study also highlighted the role of the blood-brain barrier (BBB). The BBB is a highly selective semipermeable border that prevents harmful solutes in the blood from entering the central nervous system. Using electron microscopes, the team observed large breaks in the blood vessels of the mice lacking CSE. This compromise of the BBB is a known hallmark of Alzheimer’s, as it allows toxins to enter the brain and prevents the efficient clearance of metabolic waste.
Sunil Jamuna Tripathi, co-first author and researcher in Paul’s lab, emphasized the multifaceted nature of the damage. "The mice lacking CSE were compromised at multiple levels," Tripathi said. "From DNA damage to structural breaks in the blood-brain barrier, the symptoms we observed correlated directly with what we see in human Alzheimer’s patients."
Broader Implications for Alzheimer’s Therapy
The implications of this research are profound, particularly given the current state of Alzheimer’s treatment. For decades, pharmaceutical companies have focused almost exclusively on the "amyloid hypothesis"—the idea that clearing amyloid-beta plaques from the brain would stop the disease. However, drugs targeting these plaques have often shown modest results or significant side effects.
The Johns Hopkins study suggests a different approach: metabolic and structural fortification. By targeting the CSE enzyme or finding ways to safely deliver hydrogen sulfide precursors, clinicians might be able to protect neurons before they are destroyed by plaques.
Alzheimer’s disease currently affects more than 6 million people in the United States, a number that the Centers for Disease Control and Prevention (CDC) expects to nearly triple by 2050 as the population ages. The economic burden is equally staggering, with care costs estimated in the hundreds of billions of dollars annually.
"This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," said Solomon Snyder, who co-authored the study. While Snyder retired from the faculty in 2023, his involvement in this study underscores the continuity of the research program he helped build.
Collaborative Effort and Future Directions
The research was a massive undertaking, involving contributors from Case Western University, the Leibniz Institute for Analytical Sciences in Germany, Hollings Cancer Center, the Medical University of South Carolina, and West Virginia University School of Medicine. This interdisciplinary approach was necessary to bridge the gap between chemical analysis, behavioral psychology, and advanced imaging.
Funding for the study came from a diverse array of sources, including the Department of Defense and the Department of Veterans Affairs. This highlights the growing concern over brain health within the veteran community, where traumatic brain injuries and environmental exposures may increase the risk of neurodegenerative diseases.
The next step for the Johns Hopkins team will be to investigate whether pharmacological interventions can mimic the effects of CSE. This involves developing compounds that can cross the blood-brain barrier and release hydrogen sulfide at the extremely low, consistent levels required for health without reaching toxic thresholds.
As the scientific community shifts its focus toward more complex, multi-systemic explanations for Alzheimer’s, the role of gasotransmitters like hydrogen sulfide is likely to move to the forefront. By moving beyond simple protein aggregates and looking at the fundamental metabolic health of brain cells, researchers like Paul and Snyder are opening doors to a new generation of therapies that may finally offer hope to millions.