Apex Dental Sleep Lab Secures Medicare Approval for Kava Herbst 3D Marking a Significant Shift Toward Digital Resin Manufacturing in Sleep Medicine
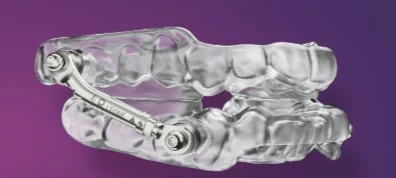
The landscape of obstructive sleep apnea treatment has reached a new technological milestone as Apex Dental Sleep Lab officially received Medicare approval for its Kava Herbst 3D oral appliance. This custom-fabricated device, which has previously secured FDA 510(k) clearance for the treatment of mild to moderate obstructive sleep apnea in adults, represents a departure from traditional manufacturing methodologies. By receiving Pricing, Data Analysis, and Coding (PDAC) verification under the Healthcare Common Procedure Coding System (HCPCS) code E0486, the Kava Herbst 3D is now positioned as a reimbursable solution for a vast demographic of patients relying on federal healthcare programs. This approval marks a critical juncture for the dental sleep industry, which has long struggled with the balancing act of high-precision manufacturing and the economic realities of declining reimbursement rates.
A Technological Evolution in Oral Appliance Therapy
For decades, the standard of care for obstructive sleep apnea (OSA) patients who are intolerant of Continuous Positive Airway Pressure (CPAP) therapy has been the Mandibular Advancement Device (MAD). Among these, the Herbst-style appliance is perhaps the most recognizable, utilizing telescoping arms to hold the lower jaw in a protruded position, thereby keeping the airway open during sleep. Traditionally, these devices were handcrafted in dental laboratories using cold-cure or heat-cured acrylics. In more recent years, the industry moved toward subtractive manufacturing, where appliances are milled from solid blocks of PMMA (polymethyl methacrylate), or additive manufacturing using Selective Laser Sintering (SLS) with nylon powders.
The Kava Herbst 3D introduces a third path: high-performance dental resin 3D printing. According to Sonnie Bocala, president of Apex Dental Sleep Lab, the move to resin-based additive manufacturing addresses several long-standing bottlenecks in the dental lab workflow. Unlike nylon SLS printing, which can result in a porous surface that is difficult to clean, or milling, which results in significant material waste, 3D resin printing allows for a high degree of precision and surface smoothness. This fully digital approach ensures that every device produced is an exact replica of the digital design, offering a level of repeatability that was previously unattainable in manual or analog workflows.
Navigating the Economic Pressures of Modern Dentistry
The impetus for the development of the Kava Herbst 3D was not merely technological curiosity but economic necessity. The dental sleep medicine sector is currently facing a "perfect storm" of rising overhead costs and stagnating insurance reimbursements. As Medicare and private insurers compress the margins for dental providers, the cost of the physical appliance becomes a primary factor in whether a practice can afford to offer sleep services.
"This project was really born out of necessity from patients, dentists, and dental labs," Bocala explained in a statement regarding the Medicare approval. He noted that as reimbursement continues to tighten, dentists are forced to become more price-sensitive regarding the appliances they select. Simultaneously, dental laboratories are grappling with the rising costs of raw materials and skilled labor. By utilizing 3D resin printing, Apex Dental Sleep Lab can scale production more efficiently, reducing the labor-intensive steps associated with traditional Herbst appliances while maintaining the durability required for a device that must withstand the forces of bruxism (teeth grinding) and nightly wear over several years.
The scalability of the Kava Herbst 3D is a central pillar of its market strategy. In a traditional lab setting, increasing production often requires hiring more technicians and expanding physical floor space for workbenches. In a digital resin-printing environment, scaling up is often as simple as adding more printing units and optimizing the digital nesting of parts. This efficiency is expected to pass savings down the value chain, making oral appliance therapy (OAT) a more sustainable clinical offering for dental practices.
Regulatory Rigor and the PDAC Verification Process
The journey to Medicare approval is a rigorous one, requiring more than just a functional design. To qualify for the HCPCS code E0486, an oral appliance must meet a strict set of criteria established by the Centers for Medicare & Medicaid Services (CMS). These requirements include being custom-fabricated for the individual patient, being adjustable (titratable) to allow for the fine-tuning of mandibular advancement, and being constructed from materials that ensure long-term clinical efficacy.
The Kava Herbst 3D underwent a formal review by Palmetto GBA, the Medicare Administrative Contractor (MAC) responsible for PDAC coding. This review confirmed that the 3D-printed resin construction met all the structural and functional requirements of the E0486 classification. This classification is vital because it provides a clear pathway for Medicare beneficiaries—many of whom are in the age bracket most frequently diagnosed with OSA—to access treatment without prohibitive out-of-pocket costs.
"From a reimbursement standpoint, having a device properly classified under E0486 is critical," Bocala stated. He emphasized that the goal was to align innovation with a structure that supports both access to care for the patient and financial sustainability for the provider. Without the PDAC verification, even the most innovative device would struggle to find a foothold in a market where Medicare serves as a primary payer.
The Clinical Impact of Obstructive Sleep Apnea
To understand the significance of this approval, one must look at the broader context of the sleep apnea epidemic. It is estimated that nearly 30 million adults in the United States suffer from obstructive sleep apnea, a condition characterized by repeated interruptions in breathing during sleep. If left untreated, OSA is linked to a host of severe health complications, including hypertension, Type 2 diabetes, stroke, and heart failure.
While CPAP remains the "gold standard" for treatment, patient compliance remains a significant hurdle. Studies suggest that between 30% and 50% of patients either stop using their CPAP machines or fail to use them for the minimum required hours per night. This "compliance gap" is where oral appliances like the Kava Herbst 3D play a life-saving role. Because they are small, silent, and do not require a mask or power source, patients often find them much easier to tolerate, leading to higher nightly usage rates and better overall health outcomes.
The Kava Herbst 3D, by being manufactured in the United States using advanced resins, offers a biocompatible and durable alternative to traditional materials. The precision of the 3D print ensures a tighter, more comfortable fit, which further enhances patient compliance. When a device fits well and is easy to maintain, the patient is more likely to wear it consistently, thereby reducing the long-term systemic risks associated with untreated apnea.
Chronology of Development and Future Implications
The path to the current Medicare approval follows a clear timeline of regulatory and developmental milestones:
- Initial R&D: Apex Dental Sleep Lab began exploring resin-based additive manufacturing as a solution to the limitations of nylon and milled materials.
- FDA Clearance: The device received its FDA 510(k) clearance (K182661), certifying it as safe and effective for the treatment of mild to moderate OSA and snoring.
- Manufacturing Optimization: The lab refined the digital workflow, focusing on high-performance dental resins that could withstand the mechanical stresses of a Herbst-style hinge.
- PDAC Submission: A formal application was made to Palmetto GBA for coding verification.
- Medicare Approval: The Kava Herbst 3D was officially assigned HCPCS code E0486, opening the door for Medicare reimbursement.
Looking forward, the success of the Kava Herbst 3D is likely to catalyze a broader shift in the dental lab industry. As resin technology continues to evolve, we may see a decline in the use of traditional acrylics and even milled PMMA. The ability to "print" complex geometries with integrated features—such as the hardware attachments for the Herbst arms—reduces the risk of mechanical failure and streamlines the repair process. If a patient loses or breaks their device, the lab can simply pull up the digital file and print a replacement, ensuring minimal interruption in the patient’s therapy.
Industry Reaction and Analysis
The industry’s reaction to the Kava Herbst 3D’s approval has been largely positive, particularly among dental sleep practitioners who have been searching for more cost-effective ways to manage their Medicare patient populations. Financial analysts in the healthcare space note that the move toward digital resin printing reflects a broader trend in medical device manufacturing: the move away from "craftsmanship" toward "data-driven production."
By removing the variability of human error inherent in analog lab work, Apex Dental Sleep Lab is essentially "de-risking" the appliance for the dentist. A more predictable fit means fewer "chairside" adjustments, saving the dentist valuable time and improving the overall patient experience. Furthermore, the use of resin allows for a more aesthetic, translucent appearance, which, while secondary to clinical function, is a significant factor in patient acceptance.
In conclusion, the Medicare approval of the Kava Herbst 3D is more than a regulatory update; it is a signal of the future of dental sleep medicine. By leveraging 3D printing to address the dual challenges of manufacturing precision and economic viability, Apex Dental Sleep Lab has provided a blueprint for how medical technology can adapt to the changing landscape of healthcare reimbursement. As more providers adopt these digital solutions, the ultimate winners will be the millions of sleep apnea patients who will have greater access to effective, comfortable, and affordable treatment options.








