Breakthrough Gene Therapy Restores Hearing in Patients with Congenital Deafness Through Successful OTOF Gene Restoration
In a landmark development for regenerative medicine and audiology, a clinical trial led by researchers from the Karolinska Institutet in Sweden and several prominent medical institutions in China has demonstrated that gene therapy can significantly and safely restore hearing in individuals suffering from profound congenital deafness. The study, published in the prestigious journal Nature Medicine, represents a pivotal shift in how genetic sensory disorders are treated, moving from mechanical compensation via cochlear implants to biological restoration of the auditory pathway.
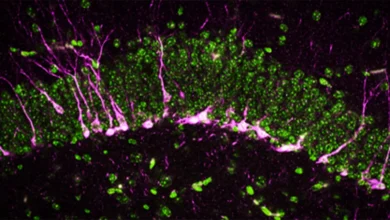
The trial focused on ten patients ranging in age from 1 to 24, all of whom were born with a specific genetic mutation in the OTOF gene. This condition, known as OTOF-related deafness, prevents the inner ear from transmitting sound signals to the brain, despite the physical structure of the ear often remaining intact. By utilizing a viral vector to deliver a functional copy of the missing gene directly into the cochlea, the research team achieved measurable hearing improvements in every participant. The success of the trial, particularly its inclusion of older children and adults, challenges previous assumptions about the window of opportunity for treating congenital sensory loss.
Understanding the Genetic Mechanism of OTOF-Related Deafness
To appreciate the significance of this breakthrough, it is necessary to understand the biological role of the OTOF gene. This gene is responsible for producing the protein otoferlin, which acts as a molecular "messenger" within the inner hair cells of the cochlea. When sound waves enter the ear and vibrate these hair cells, otoferlin facilitates the release of neurotransmitters across the synapse to the auditory nerve. Without sufficient otoferlin, the chain of communication is broken; the ear "hears" the vibration, but the brain never receives the signal.
Congenital deafness affects approximately one to two out of every 1,000 newborns worldwide, with genetic factors accounting for more than 50% of these cases. OTOF mutations are a relatively common cause of non-syndromic hearing loss, particularly in certain populations. Until now, the only effective intervention for these patients was the surgical implantation of a cochlear implant—an electronic device that bypasses the damaged parts of the ear to stimulate the auditory nerve directly. While cochlear implants are highly effective, they do not restore natural hearing and often struggle to convey the nuances of music or speech in noisy environments.
The Therapeutic Intervention: A Precision Delivery System
The treatment utilized in the study involved a sophisticated delivery mechanism known as an adeno-associated virus (AAV) vector. Because the OTOF gene is too large to fit inside a single AAV shell, researchers employed a "dual-vector" approach. The gene was split into two parts, each carried by a separate viral vector. Once injected into the patient’s ear, these two parts recombine within the target cells to form the complete, functional OTOF gene, which then begins producing the necessary otoferlin protein.
The procedure itself is minimally invasive compared to traditional ear surgeries. Under general anesthesia, surgeons administered a single injection of the gene therapy through the round window membrane—a thin tissue located at the base of the cochlea. This allows the viral vectors to enter the fluid-filled chambers of the inner ear and reach the inner hair cells.
Chronology of the Clinical Trial and Patient Progress
The trial was conducted across five hospitals in China, involving a multidisciplinary team of surgeons, geneticists, and audiologists. The timeline of the study provides a clear picture of the rapid efficacy of the treatment:
- Baseline Assessment: Prior to treatment, all ten participants were classified as having profound hearing loss, with an average auditory threshold of 106 decibels (dB). For context, 106 dB is equivalent to the sound of a power saw or a motorcycle engine at close range; participants could not hear normal speech or environmental sounds.
- Initial Post-Injection Period (0–4 Weeks): Within the first month following the single injection, most patients began to show the first signs of sound perception. Audiometry tests indicated that the hair cells were successfully producing otoferlin and initiating signal transmission.
- Intermediate Follow-up (4 Months): By the four-month mark, improvements became life-changing. A notable case involved a seven-year-old girl who had previously relied entirely on sign language and visual cues. Within 16 weeks of treatment, her hearing had improved to the point where she could engage in fluid, everyday conversations with her mother.
- Final Assessment (6–12 Months): At the six-month milestone, all ten participants showed significant hearing gains. The average auditory threshold across the group dropped from 106 dB to 52 dB. A level of 52 dB is categorized as mild-to-moderate hearing loss, allowing individuals to hear and understand speech in most settings without the aid of electronic devices.
Analyzing the Data: Age and Efficacy
One of the most critical findings of the study was the correlation between the age of the patient and the degree of recovery. While all participants improved, the younger children—specifically those between the ages of five and eight—exhibited the most dramatic gains.
This phenomenon is attributed to "neuroplasticity." The brain’s auditory cortex requires stimulation during early developmental windows to learn how to interpret sound signals. In older patients, who had lived for decades in silence, the brain had to "relearn" how to process the sudden influx of auditory data. However, the fact that a 24-year-old participant showed meaningful improvement is a major scientific revelation. It suggests that the auditory system retains a level of plasticity much later into adulthood than previously thought, opening the door for treating older teenagers and young adults who were previously considered ineligible for such biological interventions.
Safety Profile and Side Effects
Safety was a primary endpoint of the study, and the results were overwhelmingly positive. The gene therapy was well-tolerated by all ten patients. Unlike traditional pharmaceutical interventions that can have systemic side effects, this localized injection limited the presence of the viral vector primarily to the inner ear.
The most common side effect observed was a temporary decrease in neutrophils, a type of white blood cell that helps the body fight infections. This was likely an immune response to the viral vector and resolved on its own without medical intervention. No serious adverse events, such as permanent vestibular (balance) issues or systemic toxicity, were reported during the follow-up period, which lasted up to a year for some participants.
Official Responses and Expert Insights
Maoli Duan, a consultant and docent at the Department of Clinical Science, Intervention and Technology at the Karolinska Institutet, and a corresponding author of the study, emphasized the transformative nature of these results. "This is a huge step forward in the genetic treatment of deafness, one that can be life-changing for children and adults," Dr. Duan stated. He noted that while previous smaller studies in China had shown promise in toddlers, this trial’s success in older populations is what sets it apart.
"Hearing was greatly improved in many of the participants, which can have a profound effect on their life quality," Duan added. "We will now be following these patients to see how lasting the effect is. The durability of the gene expression is a key question for the next phase of research."
The medical community has reacted with cautious optimism. Independent audiologists note that while a jump from 106 dB to 52 dB is revolutionary, it does not represent "perfect" hearing. However, for a child born into total silence, the ability to hear a parent’s voice or a car horn is the difference between isolation and integration into a hearing society.
Institutional Collaboration and Funding Disclosures
The research was a massive collaborative effort, involving institutions such as Zhongda Hospital at Southeast University in Nanjing, China. The study was supported by various Chinese national research programs and Otovia Therapeutics Inc., the biotechnology firm responsible for developing the specific gene therapy used in the trial.
The involvement of Otovia Therapeutics is notable, as many of the study’s researchers are also employees or shareholders in the company. The published paper in Nature Medicine includes a comprehensive list of these disclosures to ensure transparency regarding potential conflicts of interest. This public-private partnership model is becoming increasingly common in the fast-moving field of gene therapy, where the high costs of viral vector production require significant corporate investment.
Broader Implications for the Future of Audiology
The success of the OTOF trial is being viewed as a "proof of concept" for the entire field of genetic hearing restoration. Researchers are already looking toward more common genetic causes of deafness.
"OTOF is just the beginning," Dr. Duan remarked. "We and other researchers are expanding our work to other, more common genes that cause deafness, such as GJB2 and TMC1."
The GJB2 gene, which encodes the protein Connexin 26, is the most common cause of genetic deafness in many parts of the world. However, treating GJB2 is significantly more complex than OTOF. While OTOF mutations primarily affect signaling, GJB2 mutations can affect the physical development and survival of cells within the cochlea. This means that gene therapy for GJB2 might need to be administered in utero or very shortly after birth to be effective. Nevertheless, the OTOF results provide the framework and the surgical techniques necessary to pursue these more challenging targets.
The Economic and Social Impact of Gene Therapy
Beyond the clinical results, the success of this trial has significant socio-economic implications. Congenital deafness imposes a high lifetime cost on healthcare systems and families, including the costs of specialized education, speech therapy, and the maintenance of cochlear implants, which often require hardware upgrades and battery replacements over a lifetime.
A one-time gene therapy injection, if proven to have long-lasting effects, could offer a more cost-effective solution in the long run. Furthermore, the psychological impact of restoring "biological" hearing—allowing for better pitch perception and musical appreciation—cannot be overstated. It moves the needle from "managing" a disability to potentially "curing" the underlying biological deficit.
As the global medical community watches the long-term progress of these ten patients, the focus will shift toward larger-scale Phase III clinical trials and the regulatory hurdles of bringing such a therapy to the global market. For now, the study stands as a beacon of hope for millions of families affected by genetic hearing loss, proving that the silent world of congenital deafness may one day be a thing of the past.